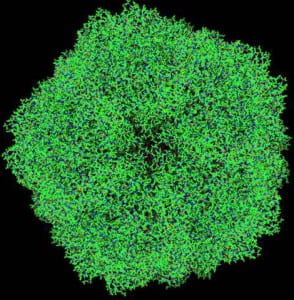
Upper part of the projected structure of satellite tobacco necrosis virus (STNV). Structural data from Protein Data Bank entry 2BUK. There are some 90,000 atoms in the entire (symmetry-expanded) capsid. Note the 5-fold axis of symmetry, typical of an icosahedral structure.
Viruses are the cause of the common cold as well as many other more dreaded diseases.
Viruses occupy the borderline between the living and the non-living. They generally consist of just two components – the genetic instructions, encoded in DNA or RNA for their own replication, and a protein coat enclosing the genetic material. Since the coat itself needs to be formed from the genetic instructions contained within the limited volume inside itself, these instructions usually code for only a small fraction of the material of th the entire coat. Consequently, as Caspar and Klug have pointed out, there are only two basic efficient designs for virus coats, icosahedral shells or helical tubes. In an icosahedral shell, for instance, the genetic material needs to code for just the asymmetric unit, a factor of at most 60 smaller than the entire material of the protein coat.
Viruses generally lack the machinery for implementing even the limited instructions of their genes. They cause diseases when they take over the protein making machinery of the cells of plants or animals and subvert the normal functions of the cells towards their own ends.
The cure for many virus-borne diseases often begins with the identification of the viruses. Even within the basic icosahedral or helical designs, there exist many variations. A picture gallery of icosahedral viruses alone may be found at e.g. the VIPERdb database.
The structures of these viruses are usually found by cryo electron microscopy, or where they can be crystallized, by x-ray crystallography. One problem with cryo electron microscopy is that electrons have limited pentration depth, and the method may be limited for the larger viruses.
A method that combines the penetrating power of x-rays with an absence of a need for crystallization is provided by the novel capabilities afforded by the x-ray free electron laser (XFEL), many of which are in construction in different countries around the world, and one of which, the Linac Coherent Light Source (LCLS), is already operational at Stanford in California.
This entraordinary machine, built at the cost of hundreds of millions of dollars, produces x-rays some billion times brighter than any existing x-ray source! This allows for the possibility of the measurement of the scattering from individual nanoparticles as small as viruses.
Methods have been devised for injecting individual proteins for scattering by the XFEL beam. Diffraction patterns from individual virus particles can be measured at a rate of up to 100 per second, and thus a large number, even up to a million can be measured in an individual experiment. A problem is that the orientations of the particles are usually unknown and that the diffraction patterns may have very low detected photon counts per pixel. Theoretical methods have been proposed for overcoming both problems, one of which has been suggested by ourselves.
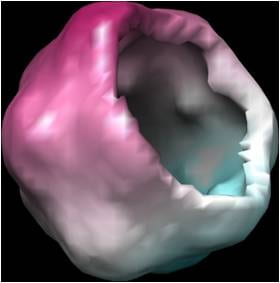
Reconstructed image of satellite tobacco necrosis virus (STNV) from synthetic single-molecule diffraction data. The image has been sliced computationally to reveal the hollow nature of the icosahedral capsid.
We have recently performed simulations which suggest that the structure of an icosahedral virus may be reconstructed by such a method from data in such measured diffraction patterns.