Projected structure of the K-channel protein from structural data in the Protein Data Bank. In nature, this protein straddles a membrane and provides a channel for the exchange of K ions between a biological cell and its environment. Note the central pore in this projection, parallel to the ion channel (and perpendicular to the membrane when embedded), as well as the X-shaped feature denuded of atoms.
Although the amino-acid sequences of more than 3/4 million proteins have now been ermined, the 3D molecular structures of less than 6 percent of these have been determined, largely by x-ray crystallography. Only 1 percent of the elucidated protein structures are of membrane proteins, despite the fact that the latter are the targets of 70 percent of today’s pharmaceutical drugs. The main bottleneck is the fact that membrane proteins are designed to be located in the water-less environment of a bilipid membrane, and thus contain hydrophobic surfaces which are generally resistant to crystallization within the hydrated environment of a crystal.
One of the advantages of a crystal is that a very large number of molecules are arranged in identical orientations, and so all that is required is that an aggregate flux reaches a minimum value for an easily detected signal of scattered x-rays. The incident flux each molecule may be kept below the threshold for radiation damage.
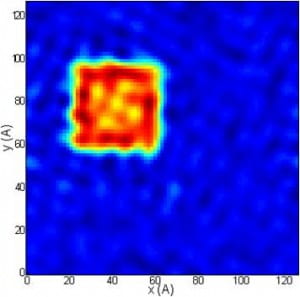
Reconstructed image of the projected structure of the K-channel protein from a simulation of a short pulse of x-rays incident on a “black lipid membrane” containg a large number of such molecules in random orientations about an axis perpendicular to the membrane. Note that the central pore and X-shaped feature of this projection has been reconstructed.
We have developed a method which can take advantage of scattering by a large number of molecules keep the x-ray dose on individual molecules smaller than the radiation damage threshold, while relaxing the requirement for crystallization. Indeed, in its first simulation for membrane proteins, its has been shown to be able to determine the projected structure of a membrane protein from a large number of copies in situ , within the hydrophobic environment of a bilipid membrane .
The basic idea has been confirmed by experiments at the Advanced Light Source (ALS) at Berkeley on a model system of metal rods (about the size of a typical virus) on a flat SiN substrate.